Arboviruses have emerged as a major global public health problem over the last 50 years, and are among the most important cause of emerging epidemic infectious diseases worldwide.
Arboviruses are maintained in complex biological cycles, typically involving a primary vertebrate host and a primary arthropod vector. An arthropod vector can be a mosquito, a flea, a sand fly, a lice, a flea, a tick or a mite. Most are zoonoses or viruses that have vertebrate animals other than humans as their principal reservoir host. These cycles exist in natural foci that are unknown until humans, their domestic animals, and other animals encroach natural habitats causing an outbreak or an epidemic (Figure 1).
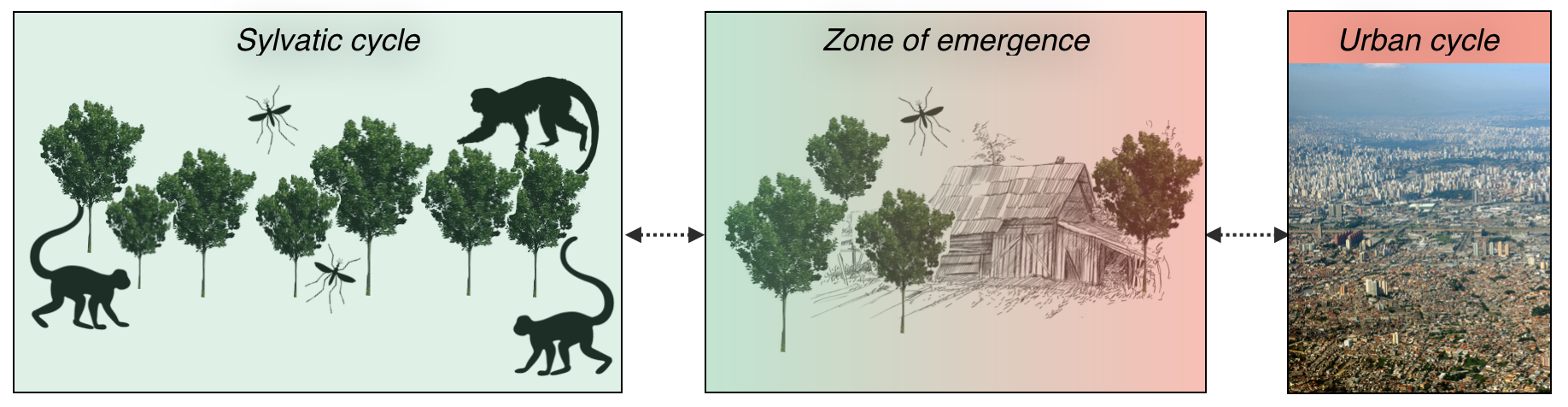
Figure 1. The biological cycle of yellow fever and Zika virus. Their cycle involves transmission in a sylvatic cycle, also known as jungle cycle, with transmission mediated by sylvatic mosquitoes (e.g. Haemagogus and Sabethes spp. for yellow fever virus in the Americas) and an urban cycle where transmission is mediated by urban mosquitoes (e.g. Aedes aegypti and Aedes albopictus). The last urban outbreak of yellow fever in Brazil was in 1928-1929 in Rio de Janeiro with 436 deaths. The early history of yellow fever in Brazil is detailed in a landmark report by Dr. Odair Franco in 1969.
Several factors are responsible for the emergence and resurgence of arbovirus diseases, including rapid population growth, human migration and illegal animal trade, unplanned and uncontrolled urbanization, human encroachment on natural disease foci such as forested areas, deforestation and land use, lack of effective mosquito control and poor health infrastructure, and climate change.
Until today, nearly 500 viruses have been registered as arboviruses, with 25% of them registered in South America, 25% in Africa. Of these 135 are known to infect humans. In general, arboviruses that infect humans cause a broad range of illness that ranges from asymptomatic infection disease to severe, chronic or fatal disease. In general, arboviruses cause three main disease symptoms: (i) systemic febrile illness, (ii) hemorrhagic fever; and (iii) invasive neurological disease.
Most arboviruses such as ZIKV and DENV cause nonspecific febrile illness during acute phase followed by full recovery. However, arthritic sequelae are frequent in CHIKV infection, and haemorrhagic manifestations occur in a significant proportion of patients with YFV. Neurological complications such as microcephaly occur in 3% of infants born from mothers infected with ZIKV. Moreover, meningoencephalitis or acute flaccid paralysis can occur upon WNV infection.
A number of viruses have adapted to human transmission in the Americas, including dengue (DENV), Zika (ZIKV), chikungunya (CHIKV), yellow fever (YFV), West Nile Virus (WNV), Venezuelan Equine Encephalitis (VEEV), Mayaro virus (MAYV) and Oropouche (OROV). With the advent of novel sequencing technologies, improved diagnostic of co-circulating arboviruses has been made possible, and new arboviruses are now being discovered from a wide range of both vector and host species.
What mosquito species transmit arboviruses?
There are over 3,000 mosquito species around the world, but only a few are responsible for spreading diseases to humans. Among those, the primary mosquito species transmitting Zika, Chikungunya, Dengue and yellow fever viruses are highlighted below.
Aedes aegypti
Taxonomy
Species from the Culicidae family, genus Aedes, subgenus Stegomyia (Linnaeus, 1762).
Distribution
Aedes aegypti occupies urban areas with or without vegetation in tropical and subtropical regions, and can be found throughout all regions in Brazil.
Habits
This species bites, rests, and lays eggs both indoors and outdoors. Majorly produced in human-made containers, treeholes and bamboo internodes holding water. Most containers with water used for immature development are within or in close proximity to households.
Arbovirus transmission
Aedes aegypti is naturally found infected with DENV, ZIKV and CHIKV. The mosquito breeds indoors and us capable of biting anyone throughout the day.
Additional information
Combating Aedes aegypti by the Brazilian Ministry of Health, Aedes aegypti ECDC fact sheet and How to Control Aedes aegypti by the CDC.
Haemagogus leucocelanus
Taxonomy
Species from the Culicidae family, genus Haemagogus, subgenus Conopostegus (Dyar & Shannon, 1924).
Distribution
The species has been found throughout Brazil, Argentina and Paraguay.
Habits
The species seems to exhibit greater plasticity of habitats and blood feeding patterns than Haemagogus janthinomys. Haemagogus leucocelanus colonizes secondary and modified forest patches and usually attacks humans at lower forest strata. Species has high dispersion capacity between forest fragments, and may bite distant from the woods and even indoors.
Arbovirus transmission
Naturally found infected with YFV, Ilhéus, Maguari, Una and Wyeomyia virus (WYOV). It is the principal vector of YFV in Sao Paulo state.
Aedes albopictus
Taxonomy
Species from the Culicidae family, genus Aedes, subgenus Stegomyia (Skuse,1984). Also known as the Asian tiger mosquito.
Distribution
The species is associated with thickets and arboreal vegetation in tropical and subtropical regions, and can be found throughout all regions in Brazil. It is primarily a forest species that has become adapted to rural, suburban and urban human environments.
Habits
It is mostly an outdoor mosquito. Shows preference for treeholes and bamboo internodes with water but can also utilize human-made containers for its immature development. Utilizes water-filled containers around or further away from households.
Arbovirus transmission
Aedes albopictus is naturally found infected with DENV and CHIKV but mostly as a secondary vector.
Additional information
Haemagogus janthinomys
Taxonomy
Species from the Culicidae family, genus Haemagogus, subgenus Haemagogus (Dyar, 1921).
Distribution
The species has been found throughout Brazil, north of Argentina, Colombia, Bolivia, Ecuador, Guinas, Peru, Venezuela, Suriname and Trinidad and Tobago.
Habits
Species has high dispersion capacity between forest fragments, and may bite distant from the woods and even indoors. Haemagogus janthinomys has a diurnal activity. It feeds on a variety of host species, including birds, humans, cattle, opossums, dogs and horses.
Arbovirus transmission
Naturally found infected with YFV, Codajas, Jurara and Mayaro virus. It is the principal vector of sylvatic YFV in Brazil.
Aedes scapularis
Taxonomy
Species from the Culicidae family, genus Aedes, subgenus Stegomyia (Rondani, 1848).
Distribution
The species is found at low to moderate elevations in tropical and subtropical regions, and can be found throughout all regions in Brazil.
Habits
Females of scapularis attack humans readily, and though primarily crepuscular, the mosquito shows diurnal and nocturnal activity.
Arbovirus transmission
Aedes scapularis has been found infected with several arboviruses, including YFV, Rocio, Melão, Ilhéus, Mayaro, Venezuelan equine encephalitis viruses.
References
Segura MNO, Castro FC (2007) Atlas de Culicídeos na Amazônia Brasileira, Características especificas de insetos hematofagos da damilia Culicidae, Instituto Evandro Chagas – MS/SVS, Seção de Arbovirologia e Febres Hemorrágicas, Belém, Brasil.
Gubler D, Vasilakis N (2016) The Arboviruses: Quo Vadis? In Arboviruses – Molecular Biology, Evolution and Control, Edited by Nikos Vasilakis and Duane Gubler, Caister Academic Press.
Pauvolid-Corrêa A, Kenney JL, Couto-Lima D, Campos ZM, Schatzmayr HG, Nogueira RM, et al. (2013) Ilheus virus isolation in the Pantanal, west-central Brazil. PLoS Negl Trop Dis 7: e2318.
Arnell JH (1976) Mosquito studies (Diptera, Culicidae). XXXIII. A revision of the Scapularis Group of Aedes (Ochlerotatus). Cont Amer Entomol Inst 13: 1–144.
Wilkerson RC, Linton Y-M, Fonseca DM, Schultz TR, Prince DC, Strickman DA (2015) Making Mosquito Taxonomy Useful: A Stable Classification of Tribe Aedini that Balances Utility with Current Knowledge of Evolutionary Relationships, PLoS One, 10(7): e0133602.
Vasconcelos PFC, Travassos da Rosa APA, Dégallier N, Travassos da Rosa JFS, Pinheiro FP (1992) Clinical and eco-epidemiological situation of human arboviruses in Brazilian Amazonia. Ciência Cultura (J. Braz. Assoc. Adv. Sci.) 44: 117–124.
Cunha MS, Faria NR, Caleiro, GS, Candido DS, Hill SC, Claro IM, Charlys da Costa A, Nogueira JS, Maeda AY, Gisele da Silva F, Pereira de Souza R, Spinola R, Tubaki RM, Tironi de Menezes RM, Abade L, Mucci LF, Timenetsky MCST, Sabino E (2020) Genomic evidence of yellow fever virus in Aedes scapularis, southeastern Brazil, 2016. Acta Tropica, 205(105390).